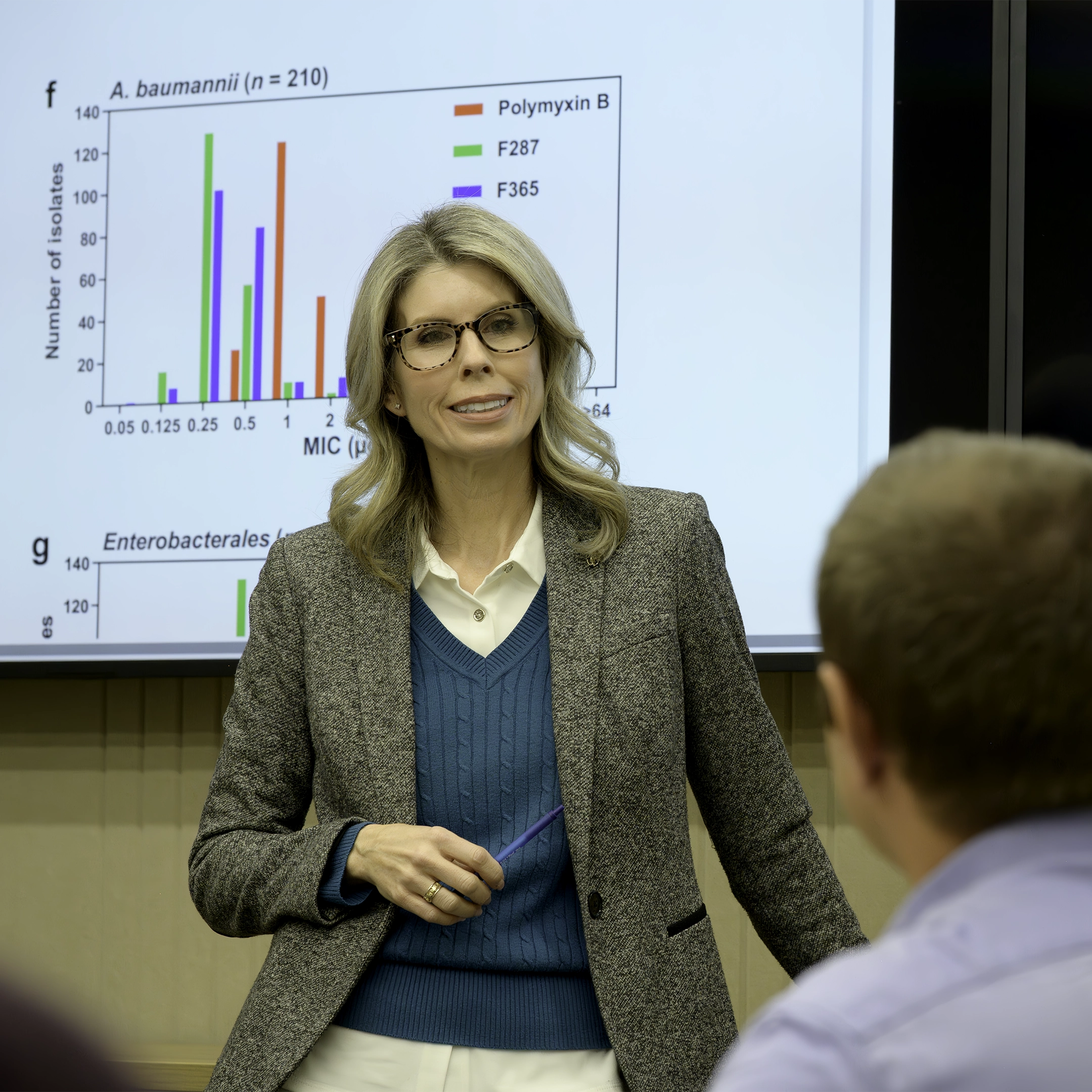
There are hundreds of known bacterial and fungal pathogens.1, 2 With antimicrobial resistance (AMR) adversely impacting healthcare worldwide, there’s an urgency to act. Meanwhile, the antibiotic R&D pipeline has continued to decline, heightening the unmet need to develop new treatments.3 Our pipeline is focused on addressing this challenge, dedicated to creating clinically proven anti-infectives.
Antibiotic Resistance: Lots of Small Problems Have Become a Big Problem
In the aggregate, antibiotic resistance is a big problem. However, from a drug discovery and innovation perspective, it is useful to think about the big problem of antibiotic resistance as many small problems, since antimicrobial-resistant pathogens often require targeted solutions. While the annual number of cases varies across pathogens, infections due to individual antibiotic-resistant pathogens considered by the U.S. Centers for Disease Control and Prevention (CDC) to be serious or urgent threats typically impact fewer than 100,000 persons per year in the U.S.4 As such, discovering and developing new antibiotics can be analogous to the challenge of finding new drugs for orphan or rare diseases. Rather than treating all antimicrobial-resistant pathogens with a single solution, we need a continuous supply of new antibiotics that target one or more pathogens with emerging resistance.